- EASY TO UNDERSTAND RESULTS – Fact sheet for patients and the official lab report will be available to you online and may be downloaded to serve as proof of your result. DxTerity developed the test and each sample is processed in our CLIA-Certified (Clinical Laboratory Improvement Amendments) and CAP Accredited (College of American Pathologists) laboratory.
If you are not sure of the importance of buying products made in the USA, please visit our main site where we make a very compelling case on the virtues of buying American products and in this case the DxTerity COVID-19 Saliva at-Home Collection Kit.
Thank you for buying American.
Register, Collect and Ship Same Day
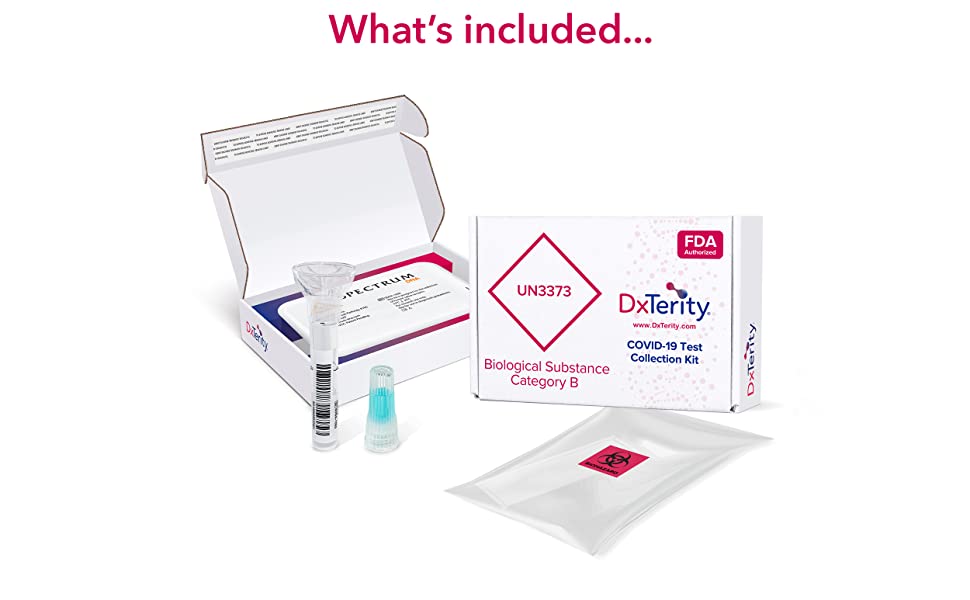
Simply register your kit, create or log into your account, complete the screening questionnaire, and type in the 14-digit Specimen ID located on your collection tube. The Specimen ID must be entered correctly to process your test. Open the kit and place all the contents on a clean, dry surface. Register your device just before you collect your sample. Your Specimen ID and the registration date and time will be electronically stamped upon registration.
Freedom is just a few steps away
Reliable COVID-19 testing utilized by over 100 U.S. corporate partners
Companies from coast to coast trust DxTerity’s COVID-19 screening program. Now available to the general public on Amazon.
DxTerity Diagnostics is a Los Angeles based CLIA licensed and CAP/ISO accredited clinical laboratory leveraging the power of genomic testing with safe and convenient from-home sample collection capabilities. DxTerity’s COVID-19 testing program has clinically validated and is EUA authorized by the FDA.
This test was developed, its performance characteristics determined, and testing performed by DxTerity Diagnostics Inc. located at 19500 S. Rancho Way, Suite 116, Rancho Dominguez, CA 90220, regulated under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42U.S.C. as qualified to perform high complexity clinical testing. This test has not been FDA cleared or approved. This test has been authorized by FDA under an Emergency Use Authorization (EUA) for use by DxTerity Diagnostics, Inc. This test has been authorized only for the qualitative detection of nucleic acid from SARS-CoV-2 virus that causes COVID-19, not for any other viruses or pathogens. This test is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or revoked sooner. Results of the test should not be used as the sole basis for patient management decisions. You cannot use this test if you are experiencing any of the following emergency warning signs for COVID-19: Trouble breathing, Persistent pain of pressure in the chest, New confusion, Inability to wake up or stay awake, Bluish lips or face – It is recommended that you immediately seek emergency care. Refund policy: the kit is not returnable once purchased and must be used within 60 days of purchase, refunds will be provided if the physician prescription is not authorized for any reason (such as for having emergency warning symptoms at sample registration) or if results are not provided due to fault of the shipment or testing process. Do not eat or drink for 30 minutes prior to collection. Samples must be registered into the portal at my.dxterity.com and returned to a FedEx collection location on the same day as collection, before the last express pick-up. Samples may not be collected on Sunday in most locations. Please see my.dxterity.com for complete terms of service and privacy policy.